Rust is easily the most cited example of metal corrosion. However, there’s no way we can expect the diverse array of heavy metals to develop identical reactions to oxidation.
To understand rust, we need to look to science. That reddish-brown rust you see on pipes, sheeting, and other metal elements is actually a chemical reaction. Rust is the compound that’s created when the core element iron is exposed to oxygen. If you passed chemistry 101, you would probably remember that a chemical change is different than a physical one. In this case, it’s iron (Fe) becoming iron oxide or ferric acid (Fe2O3), a completely novel chemical compound.
Brass, zinc, and titanium are all heavy metals with negligible amounts of iron. For this reason, they’re often known as metals that never rust. In this article, we’ll take a deeper look at each of these materials and identify if any of them are even remotely susceptible to iron oxide.
What is Rust?
Before we dig into this argument, let’s talk about iron oxide. Rust is the reddish-brown coating that forms on the surface of iron that’s been exposed to moisture. While sometimes considered a sign of indifference, rust has become an increasingly popular element of style (just look at this incredibly rusty house in Oslo).

Whether you’re looking to create or avoid rust, you need to understand what happens when iron-rich heavy metals come face-to-face with oxygen. Keep in mind that while oxygen is the key catalyst, other chemicals, including salt, bleach, and vinegar, can be used to accelerate the development of iron oxide.
Does Brass Rust?
Brass is a copper alloy that is known by the element symbol Cu. It is often combined with tin to create the metal known as bronze. Since neither copper, brass, or bronze contain iron (Fe), it’s not technically capable of developing iron oxide.
With that said, copper does develop a unique patina or tarnish when it’s been exposed to moisture. Due to their different elemental breakdowns, brass (copper and zinc) and bronze (copper and tin) undergo different aesthetic changes.

When copper or tin is exposed to water, it usually develops a blue, green, or black patina. The most noteworthy example of copper oxidation is the Statue of Liberty, a copper-coated statue that has famously turned green because of oxidation. Meanwhile, bronze can become blue, green, black, brown, or even red after being exposed to H20.
Discoloration caused by oxidation can be removed. However, most people choose to embrace this metal’s natural aged patina, especially when it comes to antiques and exterior fixtures.
Exterior brass elements tend to oxidize rapidly. The only way to prevent continuous oxidation is to coat this metal in some sort of protective lacquer, such as microcrystalline wax. Protective lacquer prevents water, salt, and other contaminants from coming in direct contact with the metal.
How to Patina Brass

Are you looking to patina brass quickly? Check out this video on how to patina brass just vinegar and salt.
What You Need:
- Vinegar
- Salt
- Container for bath, spray bottle, or brush
Step 1: Remove any lacquer or varnish using acetone.
Step 2: Soak the brass item in a mixture of vinegar and salt. We recommend a ratio of one teaspoon of salt for every 1/2 cup of vinegar. This process is surprisingly quick. In most cases, it will take you no more than 10 minutes to reach your desired color.
Ferric Acid Method

What You Need:
- Ferric Acid
- Water
- Container for soaking
You can also use diluted ferric acid (ferric chloride) to patina brass objects. You can change the color of brass from brown to black depending on the ratio of ferric acid to water that you use. However, this method is more commonly used to etch the surface of brass and other copper-based metals.
Don’t worry if you don’t like the patina. In most cases, the darkened layer can be buffed out with the help of some steel wool and a little elbow grease.
How to Remove Brass Patina
What You Need:
- Lemon juice
- Baking soda
- Brush for applying
Alternatively:
- Salt
- Vinegar
- Flour
Removing the patina from brass or bronze is easy. You can easily whip up a batch of brass cleaner with common household items. One popular recipe calls for lemon juice and baking soda. Another asks for salt, vinegar, and flour. In almost all cases, you’re going to use a combination of inexpensive and readily available acids and bases to create a non-abrasive paste that can be applied to the surface of virtually any brass object.
Does Zinc Rust?
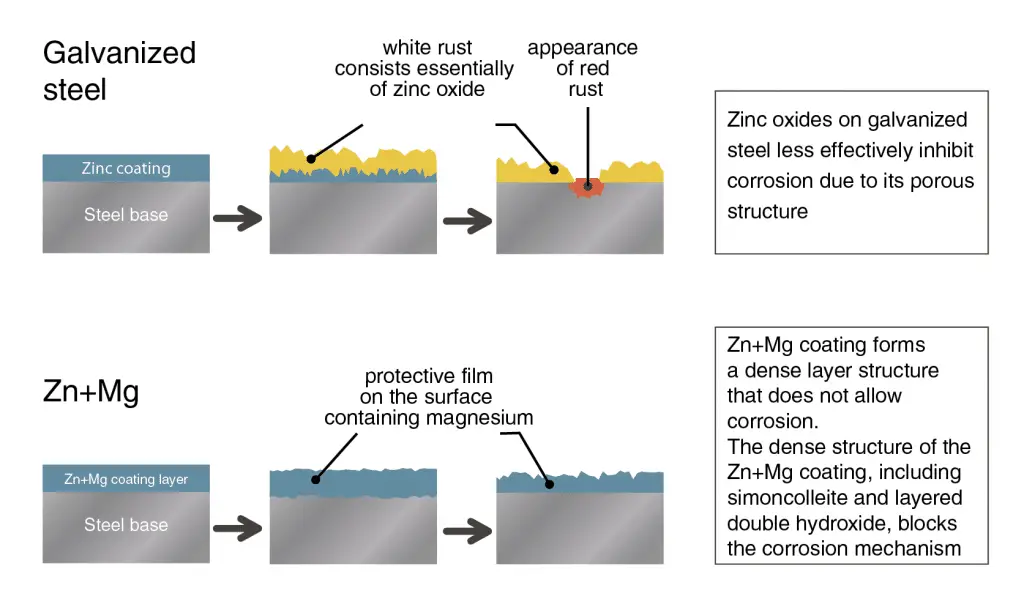
Zinc (Zn) is a silvery blue metal that is often used to galvanize steel. While zinc helps protect metals from oxidation, it is not impervious to change. In actuality, it just corrodes at a much slower rate than other heavy metals. When zinc is allowed to undergo prolonged oxygen exposure, it develops a blackish tint. We’ve got a more detailed explanation here.
Removing Zinc from Galvanized Metal
As we mentioned, galvanized metal has a thin zinc coating. This coating is supposed to protect the underlying metal (usually some sort of iron) from oxidation. While zinc coatings certainly serve their purpose, many people are looking for weathered (aka rusty) galvanized metal.
To do this, you need to remove the layer of zinc protecting the underlying steel. This can be done with the help of a number of different acid solutions, including vinegar and muriatic acid. You can soak smaller galvanized metal objects in your pick of an acidic solution. Objects that are too large for soaking will need to be sprayed or brushed with a solution.
Waiting times vary. It typically takes 24 hours for regular household vinegar to have its effect on zinc. Meanwhile, muriatic acid works faster, with visible results seen in just a few hours. You can use regular household baking soda to neutralize the acid once your metal has developed your desired patina.
How to Develop a Patina on Zinc
What You Need:
- Steel wool
- Copper sulfate
- Neutralizing agent, such as baking soda
Unweathered zinc is bright and silvery. You can age your patina to give it a darker, dull appearance. Check out this brief chemistry experiment. to see exactly how this works.

To do this, you will need steel wool and copper sulfate (CUSO4). Use the steel wool to buff the surface of the zinc. Next, submerge the object in a solution of copper sulfate (you can find this at just about any hardware store or online).
You will notice that zinc objects darken as soon as they are inserted into their blue copper sulfate baths. Zinc is easily oxidized after exposure to copper oxide.
How to Remove or Maintain a Patina from a Zinc Surface
You can use a minimally abrasive kitchen pad, dish soap, Windex, and/or water to clean zinc surfaces. Acidic compounds and household chemicals are highly likely to alter the aesthetic of this alloy.
Keep in mind that zinc is known for its ‘living finish.’ What this means is that its patina is constantly evolving. It is highly reactive and will change whenever it interacts with oxygen and acid. Therefore, each zinc item is likely to have a unique and constantly evolving pattern. So long as that pattern is not coated in a clear lacquer, you should be able to buff it out. However, in most cases, designers and architects let this material do its own thing.
Does Titanium Rust?
If you’re looking for a rust-resistant metal, then titanium may be your best bet. This iron-free heavy metal is incredibly resilient. However, with the right treatment, it will develop a thin coating of titanium oxide (TiO2). Not only does titanium oxide have the ability to change the patina of titanium, but it is also capable of protecting it against further oxidation.
A Bit About Titanium Oxide
Manufacturers may go out of their way to fabricate titanium oxide on titanium alloys. This is especially so for titanium products used in underwater and industrial applications. Titanium oxide film protects the underlying metal from a vast array of pollutants, including water, oxygen, salt, and chemicals.
What Does It Take to Generate Titanium Oxide?
Titanium oxide forms when titanium is exposed to oxygen at high temperatures. This is often done using heat anodization. You can also use OxiClean to darken titanium. Home anodization is typically used on titanium housewares. Check out this video to see scientists transform untreated titanium rods into rainbow chopsticks. As you can see, the titanium dioxide coating changes the way that light reflects off the surface of the titanium. The metallic sheen is still there. However, the surface now appears to be prismatic rather than monochrome.
Wrapping Up
As you now know, iron-based metals undergo intense chemical reactions when they are exposed to oxygen. Brass, zinc, and titanium are all susceptible to oxidation. However, since none of these heavy metals contain iron, the patinas they develop cannot be considered rust. Instead, each unique alloy formula has a reaction of its own.
Unlike traditional rust, brass, zinc, and titanium oxidation have no impact on the integrity of these metals. When iron-rich metals rust, the surface becomes flaky and weak. To put it simply, there’s no reversing the course of iron oxide. That’s not the case with the other heavy metals we’ve been discussing. Brass, zinc, and iron oxide patinas are only on the surface. In most cases, they can be removed without the underlying metal undergoing any structural changes.
Whether you’re looking to accelerate or stop your metal from oxidizing, we hope that our rundown took some of the mystique out of the process. As always, thanks for joining us for yet another exciting how-to article.



Leave a Reply